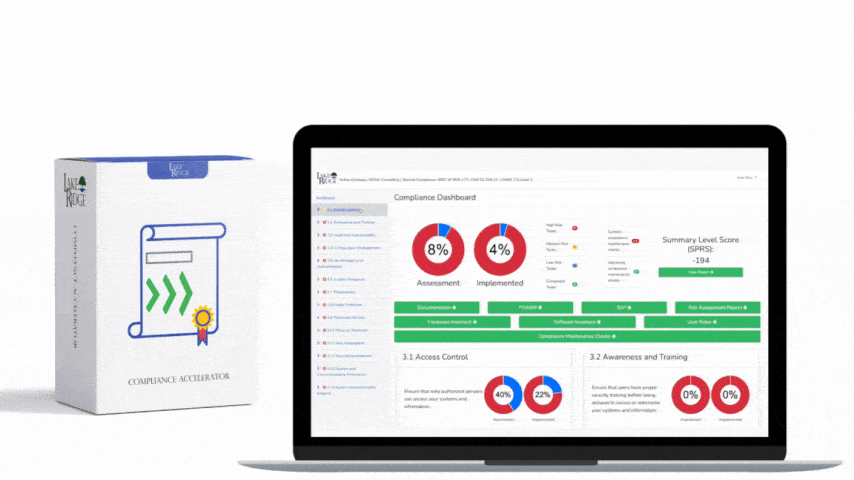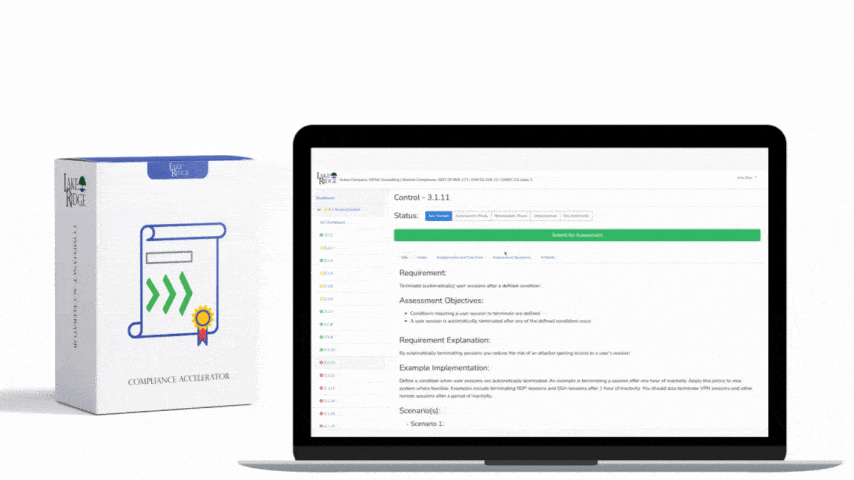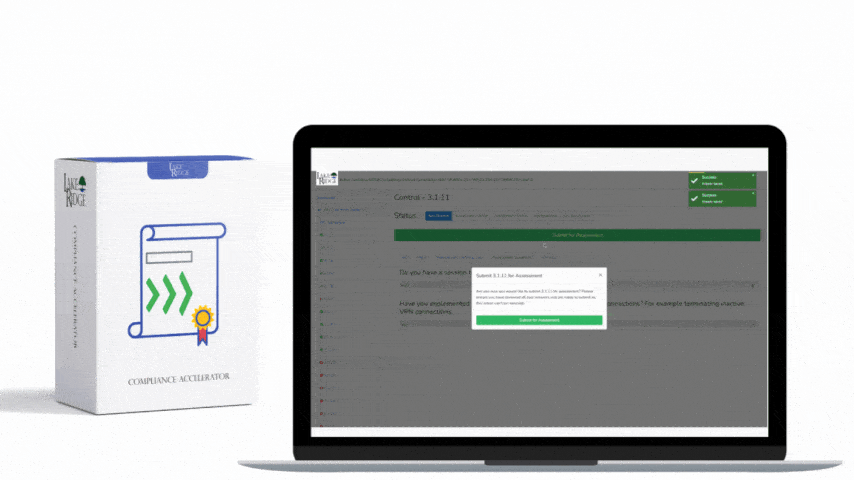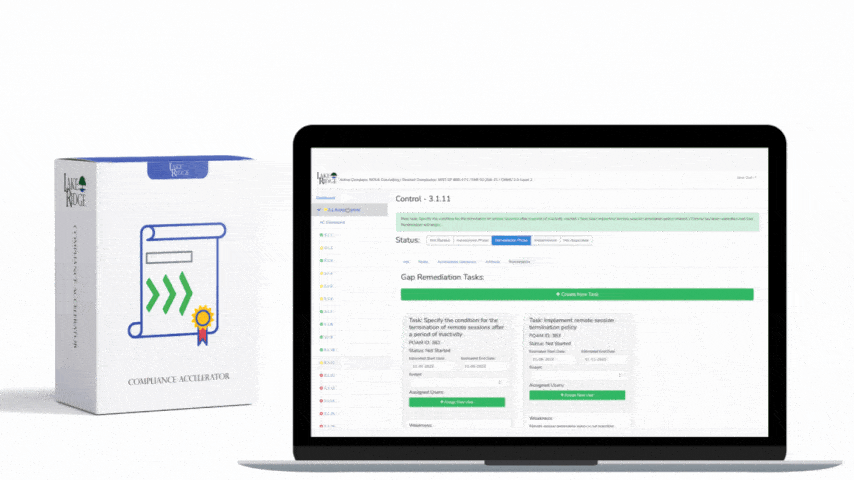
This post provides step-by-step, practical guidance for implementing risk-based controls when accepting external media that contains diagnostic and test programs, mapped to NIST SP 800-171 Rev.2 / CMMC 2.0 Level 2 (Control MA.L2-3.7.4). It explains policy, technical controls, quarantine/validation processes, monitoring, real-world small-business examples, and the types of evidence auditors will expect.
Why this control matters (risk overview)
Accepting diagnostic/test media (USB sticks, vendor recovery drives, SD cards, or vendor-provided hard drives) without controls exposes systems to malware, supply-chain tampering, and credential theft. A single infected diagnostic utility plugged into a trusted engineering workstation can pivot to production, exfiltrate Controlled Unclassified Information (CUI), or corrupt firmware on embedded devices. For contractors working under DFARS or CMMC, failure to control these vectors can lead to lost contracts, regulatory penalties, and damage to national security information.
Step 1 — Policy, roles, and a risk-based acceptance matrix
Start by creating an "External Media Acceptance Policy" that defines: allowable media types, roles (requester, approver, security reviewer), acceptance criteria tied to risk (sensitivity of systems, network segmentation), required checks (signature/hash, vendor attestation), and an exceptions process. Make the matrix risk-based: low-risk example = vendor-provided text logs reviewed on isolated VM; high-risk = firmware-update USB for a PLC — requires vendor code signing, in-person handoff, and air-gapped test bench. Record every acceptance request in your ticketing/CMDB system with a risk level and approval chain to create audit evidence.
Step 2 — Technical hardening and device control
Implement technical controls to prevent casual use of external media. On Windows: use Group Policy to disable AutoRun (Computer Configuration → Administrative Templates → Windows Components → AutoPlay Policies → Turn off AutoPlay: Enabled), apply Device Installation Restrictions by hardware ID for whitelisted devices, and employ Windows Defender Application Control (WDAC) or AppLocker to allow only signed binaries. On Linux, use USBGuard to control which USB devices can attach. Use MDM to control mobile/storage devices and consider disabling unused USB ports in BIOS/UEFI or with hardware port blockers. For Windows hashing and verification examples: use PowerShell Get-FileHash -Algorithm SHA256 C:\staging\vendor_binary.exe or on Linux sha256sum /mnt/usb/vendor_binary.bin.
Step 3 — Quarantine, scanning, and validation procedures
Never run unknown diagnostic programs on production hosts. Establish a quarantine workflow: 1) collect the media and create a bit-for-bit image (use dd or FTK Imager) to preserve chain-of-custody; 2) compute hashes (SHA-256) of original files and image; 3) scan with up-to-date AV/EDR engines and run static analysis (PEStudio, strings, YARA rules); 4) run dynamic analysis inside an isolated sandbox or an air-gapped virtual machine (Cuckoo, VirusTotal Private Monitor if allowed) that mirrors the target OS but has no access to production networks. For sensitive or proprietary code, avoid uploading to public services; use an internal sandbox or signed vendor attestations checked against a vendor certificate authority.
Step 4 — Controlled execution, network segmentation, and monitoring
If the diagnostic program must be executed, do so on a dedicated "test bench" or jump box that is logically/physically separated from production and CUI networks (dedicated VLAN, strict firewall rules). Apply least privilege to the account used to run tests, disable network shares, and restrict Internet access. Log all activity: enable verbose audit logging, collect endpoint telemetry with EDR, and forward logs to your SIEM for correlation. Use DLP to prevent accidental exfiltration. Maintain a documented chain-of-custody and attach test results, hashes, and approval tickets to the CMDB entry for the media.
Real-world small-business scenarios
Scenario A — Small manufacturing firm: a field technician receives a vendor USB containing PLC diagnostic software. Process: technician submits ticket with vendor contact → security reviews risk level (firmware update? yes) → devices are imaged and hashed on arrival → approval requires vendor code signing and an in-person handoff → software executed on isolated engineering VM in the factory VLAN with no internet and monitoring enabled → results and hashes stored in ticket. Scenario B — IT MSP: remote customer sends a diagnostic SSD containing system logs. Process: image the SSD, scan offline, redact any customer personal data before uploading to any third-party analyzer, run local static analysis, and return cleaned artifacts with a signed acceptance form.
Compliance tips, audit evidence, and best practices
Maintain the artifacts auditors want: policy documents, risk acceptance matrix, tickets/approvals, hash lists, sandbox reports, vendor attestations, and change-control records. Automate where possible: use MDM/EDR to enforce device whitelists, integrate ticketing to require security approvals before network ports are opened, and script hash computations and malware scans to reduce human error. Train field staff on the acceptance workflow and run tabletop exercises for incidents initiated by vendor media. Whenever possible, replace physical media acceptance with vendor-initiated remote diagnostics over authenticated, encrypted channels.
Summary: Implementing MA.L2-3.7.4 is a mix of policy, risk assessment, technical hardening, and disciplined operational procedures — especially for small businesses with limited staff. Use a documented, risk-based acceptance matrix, require imaging and hashing, scan and sandbox externally-provided diagnostic tools, execute only on segmented test systems, and log everything. These steps reduce malware and supply-chain risk, provide clear audit evidence, and keep your environment compliant with NIST SP 800-171 Rev.2 and CMMC 2.0 Level 2 expectations.