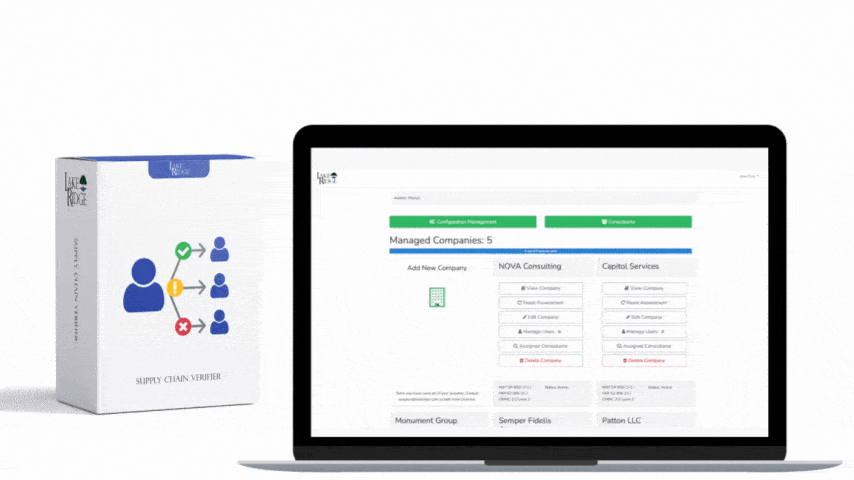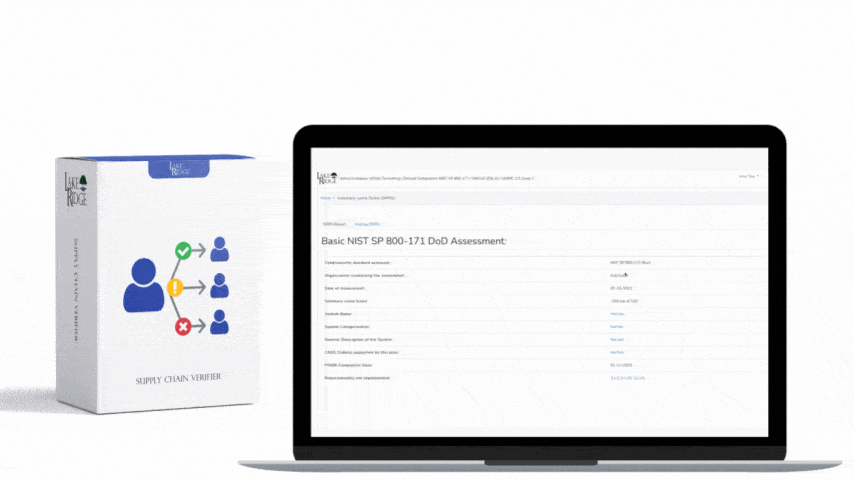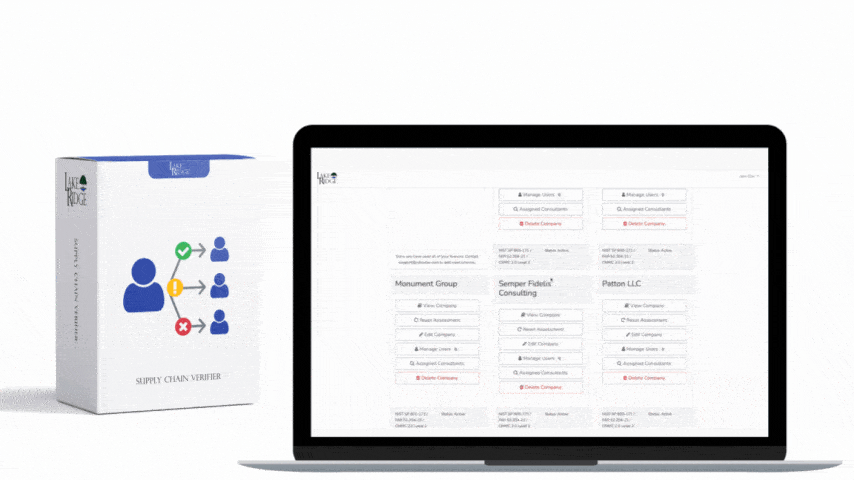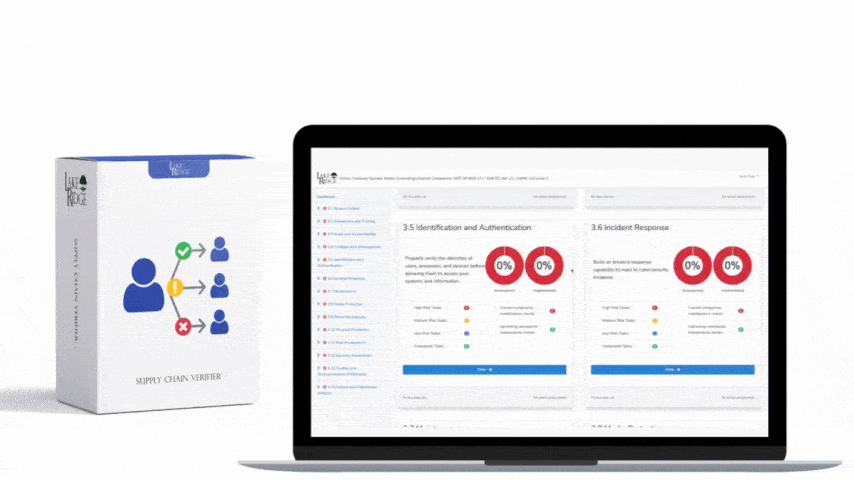
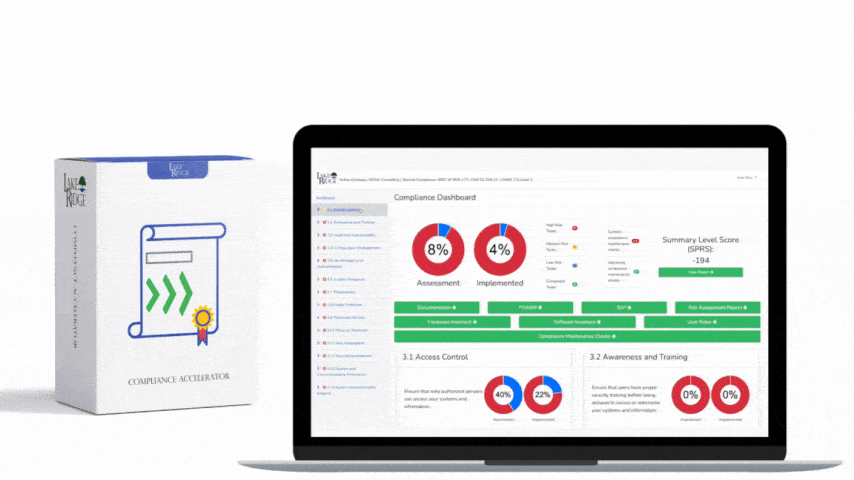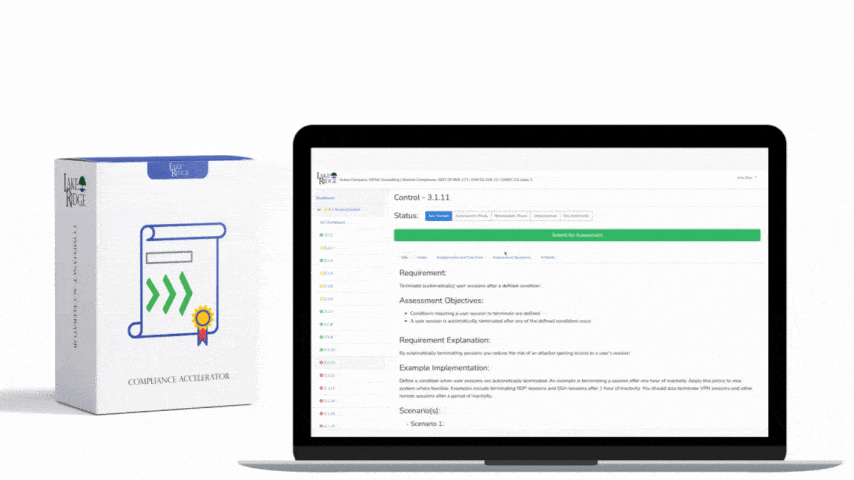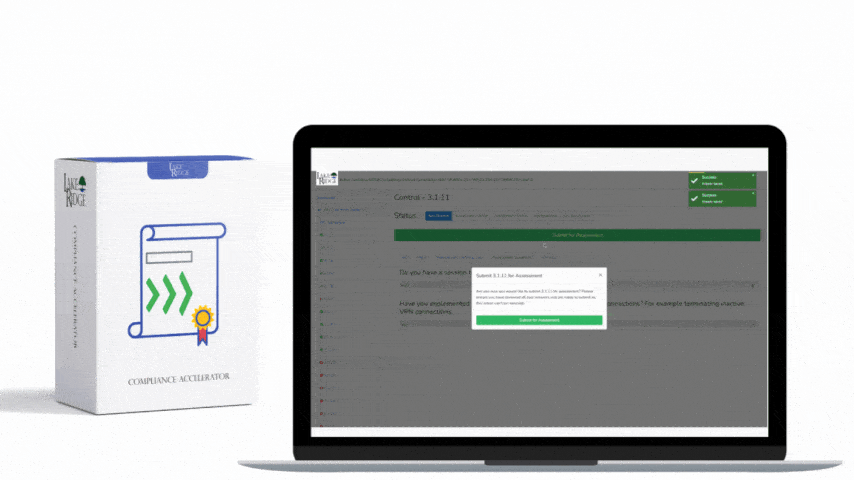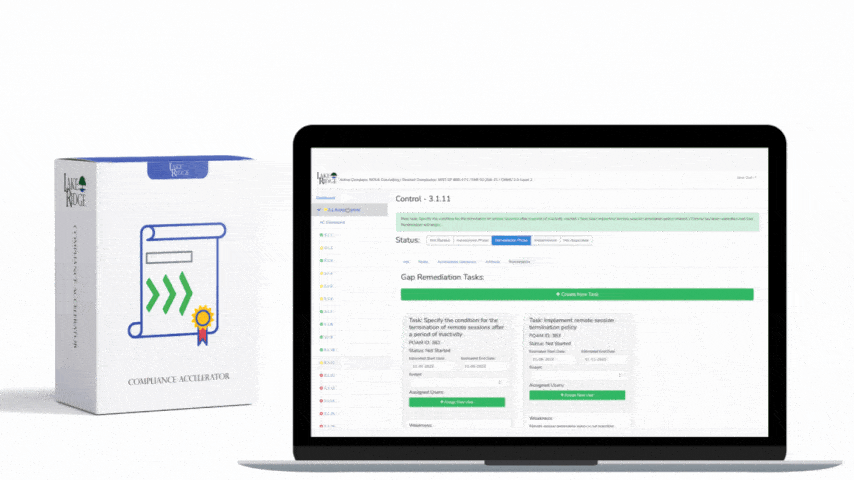
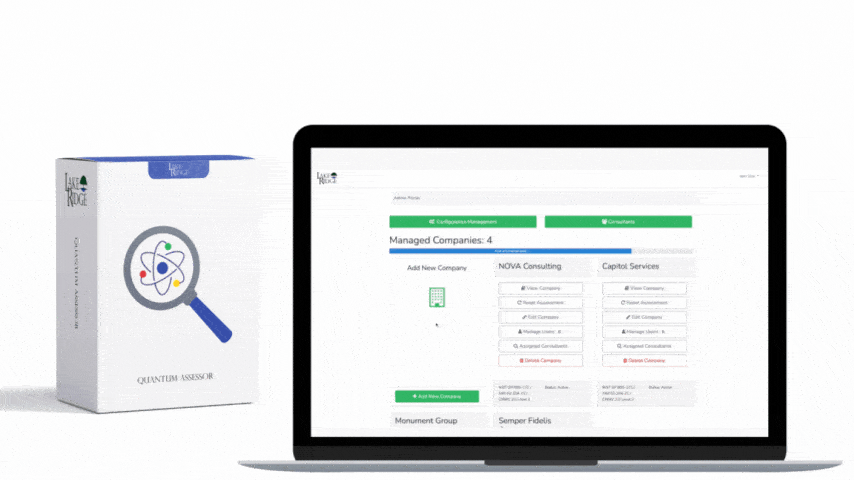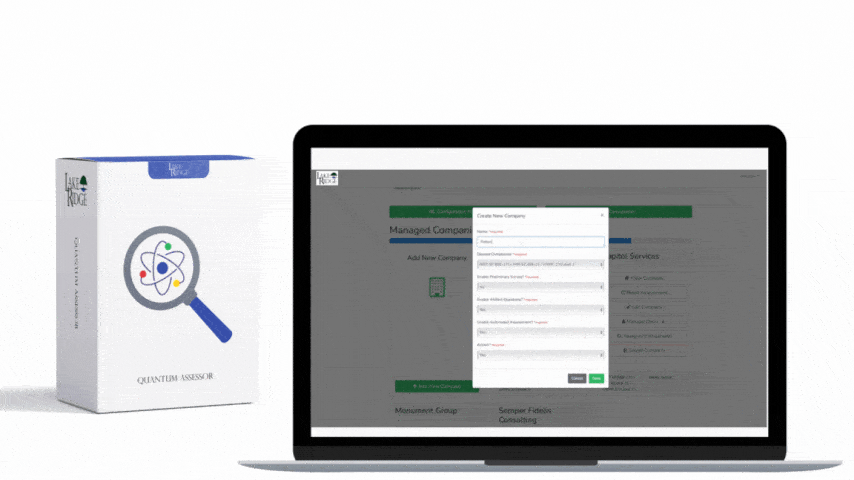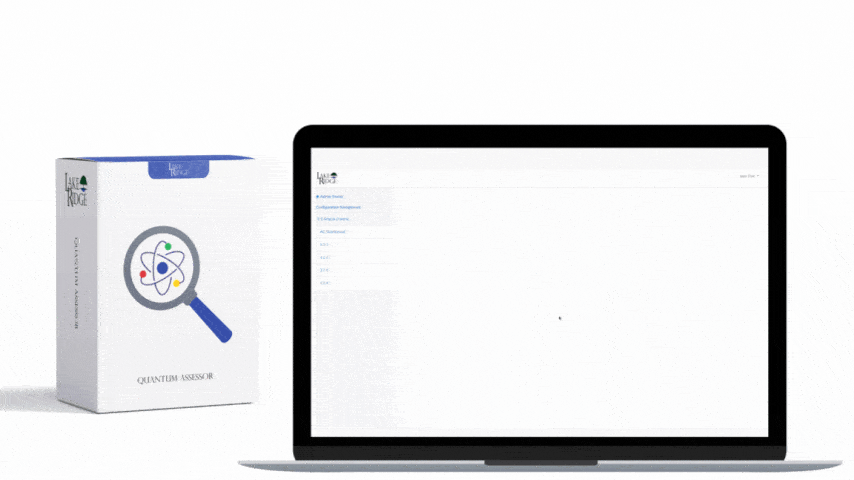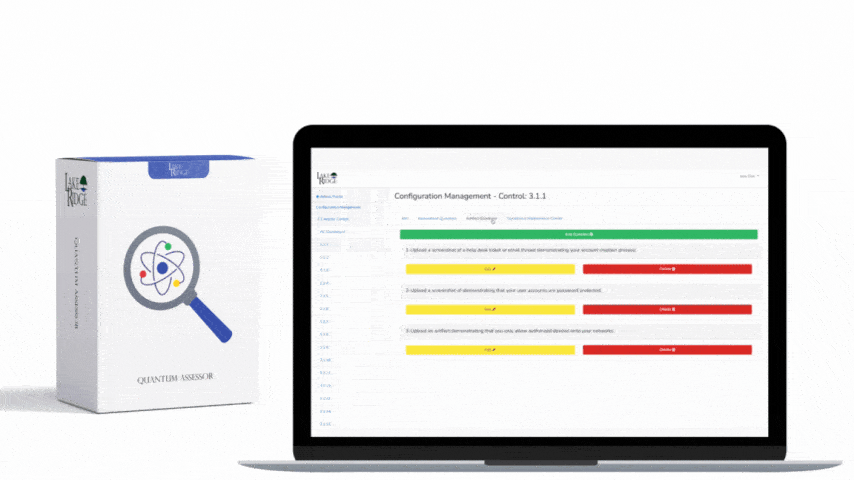
Meeting FAR 52.204-21 and CMMC 2.0 Level 1 (MP.L1-B.1.VII / Code 550) for sanitizing Federal Contract Information (FCI) is straightforward if you apply a repeatable, evidence-driven process: inventory media, map media types to approved sanitization categories (Clear, Purge, Destroy), execute the chosen method with purpose-built tools, and retain verifiable records that match your Compliance Framework documentation requirements.
Sanitization options and choosing by media type
Under recognized guidance (NIST SP 800-88 Rev. 1) sanitization is grouped into Clear (logical overwrite or crypto-erase), Purge (more robust: degauss or cryptographic rekey + wipe), and Destroy (physical destruction). For FAR / CMMC Level 1 where you handle FCI (not CUI), you still must ensure that media leaving your control cannot reveal FCI. Map media to methods: HDDs can typically be Cleared or Purged; SSDs and flash often require Purge (crypto-erase or vendor secure erase) or Destroy due to wear-leveling; magnetic tapes are good candidates for degaussing (Purge) or physical destruction; optical media and some specialized devices may need shredding or pulping.
Overwrite details — when and how to use Clear
Overwrite (Clear) remains effective for many magnetic HDDs. Practical implementation: use a vetted tool (commercial like Blancco or open-source where permitted) to write at least one full pass of pseudo-random data across all user-addressable sectors and perform a read-verify pass. For Compliance Framework evidence, capture the tool output: device serial, model, software version, start/end timestamps, pass patterns, checksum/hash of post-sanitized sectors, and operator ID. Note that for modern high-density HDDs a single-pass overwrite is generally sufficient per current guidance, but record your rationale in policy. Do not rely on overwrite for SSDs unless your policy explicitly references a vendor-supported secure-erase procedure and you can validate its effect.
Degaussing — what to require and verify
Degaussing (Purge) is appropriate for magnetic media such as hard drives and tapes but not for SSDs, USB flash, or optical media. Practical steps: procure a degausser rated to the media type (confirm areal density and required magnetic field strength), test it on a sacrificial device of the same model to prove efficacy, and record serial numbers and a degauss certificate for each device processed. Commercial degaussers will specify field strength (often in Tesla or Gauss); for compliance, demand manufacturer test reports and keep calibration logs. After degaussing, attempt a simple device re-detection to show the drive is non-functional or unreadable and include that as part of the artifact set for auditors.
Physical destroy — methods, specs, and evidence
Physical destruction is the fallback when Clear/Purge are infeasible or for media with very high risk. Options include shredding (cross-cut or industrial disintegrator), crushing/fracturing, or incineration. For small businesses: use a reputable third-party vendor with NIST/DoD-aligned particle size or NSA/DISA guidance where contractually required. Record the chain-of-custody, witness signatures, a certificate of destruction that lists device serials, and photos/video if possible. For self-performed destruction, document the procedure, equipment specs (shredder model, particle size), operator training, and post-destruction sampling to prove un recoverability.
Verification, logging, and evidence for audits
Verification is where many small businesses fail. Practical verification steps: (1) capture pre-sanitation asset records (serial number, model, FCI owner), (2) during sanitation collect machine-generated proof (overwrite logs, degauss certificates, shredding certificate), (3) perform post-sanitation validation — for overwrite, run read-after-write checks or compare sector checksums; for crypto-erase, record the key destruction event and tool output; for degauss/physical, capture vendor certificate and visual proof. Maintain these records for the contractually required retention period and make them available for FAR / CMMC evidence review. Implement a simple naming and storage convention (e.g., /compliance/media-sanitization/YYYYMMDD-assetID.pdf).
Small-business implementation example
Example scenario: a 25-person subcontractor has 20 retired laptops, 5 backup tapes, and a dozen USB drives containing FCI. Implementation: inventory (asset list + serials), classify media (HDD in laptops = overwrite or crypto-erase; tapes = degauss or destroy; USB = destroy), select tools (vendor ATA Secure Erase for laptops with SSDs or use vendor-provided crypto-erase; Blancco or a validated overwrite tool for HDDs; contract a local certified shredding vendor for USBs and tapes). Execute in a controlled event: two staff sign chain-of-custody sheets, run sanitization with automated logging, and store certificates. If an auditor inspects, you can show the asset list, sanitization logs, chain-of-custody, and certificates for each device.
Risks of not implementing adequate sanitization
Failure to sanitize FCI properly introduces legal, financial, and reputational risks: unauthorized disclosure of FCI may lead to contract termination under FAR 52.204-21, loss of future contract opportunities, monetary penalties, and expensive incident response. From a technical perspective, inadequate sanitization can lead to data remanence and successful recovery by attackers or forensic analysts. Demonstrable controls (logs, certificates, SOPs) mitigate risk and are central to passing CMMC assessments and contract reviews.
Compliance tips and best practices
Actionable tips: embed sanitization steps in your asset lifecycle (procurement → inventory → use → sanitization → disposal); standardize on vendor tools and keep versions approved in policy; use third-party vendors for destruction when you cannot internally prove effectiveness; train staff on chain-of-custody and evidence collection; perform quarterly spot-checks (for example, attempt forensic recovery on a sample of sanitized devices) and document results; ensure all sanitization activities reference your Compliance Framework mapping to FAR 52.204-21 and CMMC controls in your policies and PO/contract clauses.
Summary: Selecting and verifying sanitization methods for FCI under FAR 52.204-21 and CMMC 2.0 Level 1 is a predictable process—classify media, choose Clear/Purge/Destroy based on media type and vendor guidance, execute with validated tools or vetted vendors, and retain structured evidence (logs, certificates, chain-of-custody). For small businesses, pragmatic steps—inventory, vendor contracts, automated logging, and periodic validation—deliver compliance and dramatically reduce the risk of data leakage and contract penalties.